Safety, Tolerability and Acceptability of Long-Acting Cabotegravir (CAB LA) for the Prevention of HIV among Adolescent Males – A sub-study of HPTN 083

Study Documents - Study Details - Key Study Personnel - Study Sites - Publications
What is HPTN 083-01?
HPTN 083-01 is a clinical trial examining whether injectable cabotegravir (CAB) for PrEP (pre-exposure prophylaxis) is safe and acceptable for adolescent males [assigned male at birth – including men who have sex with men (MSM), transgender women (TGW), and gender nonconforming people].
Who is participating in HPTN 083-01?
This study will be enrolling about 50 adolescent males (assigned male at birth – including MSM, TGW, and gender nonconforming people) under the age of 18 at sites in Boston, Chicago, and Memphis.
Why is HPTN 083-01 important?
Adolescents and young adults make up a large number of new HIV infections worldwide. Although taking a daily pill for PrEP works to protect people from HIV, having to take a daily pill has proven to be difficult for some people. Long-acting injectable PrEP has the potential to protect people from HIV without having to take a daily pill.
What will happen during HPTN 083-01?
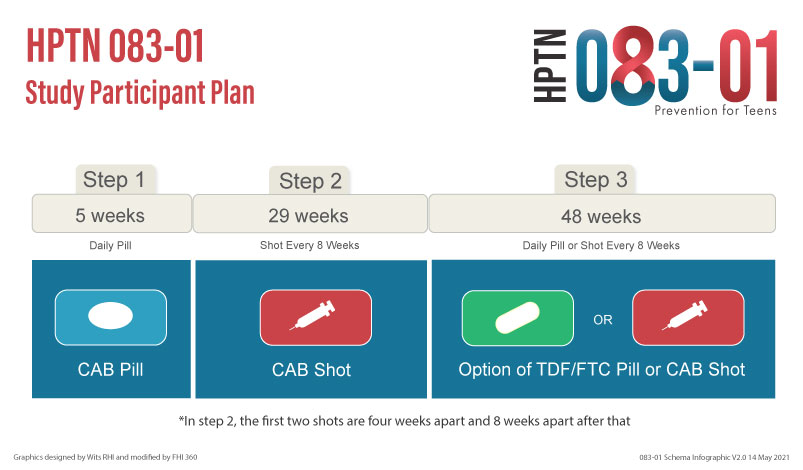
Participants will move through the study in 3 steps:
Step 1: Participants will take one CAB pill every day for five weeks
Step 2: Participants will receive a total of 5 CAB injections over 6 months
Step 3: Participants will come to the clinic for study visits quarterly and receive Truvada for PrEP for about one year.
Study Documents
Version 3.0
- HPTN 083-01 Version 3.0 - 2 July 2021
- HPTN 083-01 Summary of Changes from V2.0 to V3.0 - 2 July 2021
- HPTN 083-01 Version 3.0 LoA #1 -14 January 2022
- HPTN 083-01 Version 3.0 LoA #2 - 17 February 2022
- HPTN 083-01 Version 3.0 LoA #3 - 15 August 2022
- HPTN 083-01 Protocol 3.0 CM #1 - 18 May 2023
Version 2.0
- HPTN 083-01 Version 2.0 - 23 August 2020
- HPTN 083-01 Summary of Changes from V1.0 to V2.0 - 23 August 2020
Version 1.0
- HPTN 083-01 Version 1.0 - 3 October 2019
➤ HPTN 083-01 SSP
(Click to Expand)
HPTN 083-01 Study-Specific Procedures Summary of Changes - 8 December 2022
Section 1: Introduction
Section 2: Protocol
Section 3: Document Requirements
Section 4: Recruitment, Screening, and Enrollment
Section 5: Study Procedures Overview
Section 6: Visit Checklists
Section 7: Participant Retention
Section 8: Study Product Considerations
Section 9: Clinical Considerations
Section 10: Adverse Event Reporting and Safety Monitoring
Section 11: Laboratory and Specimen Management Procedures
Section 12: Counseling Considerations
Section 13: Data Management
Section 14: Computer Assisted Self-Interview (CASI)
Section 15: Reporting Plan
Section 16: Data Communiqués
Section 17: Qualitative
Appendix 1A: Record of Dispensation of Participant-Specific Study Product to Non-Pharmacy Staff
Appendix 1B: Record of Product Return of Participant-Specific Study Product by Non-Pharmacy Staff
Appendix II: Participant Transfer and Receipt Process within Medidata Rave
Appendix III: Guidance for the Management of “Discordant/discrepant” HIV Testing Results
Appendix IV: Study Schema
Other tools
Presentations
Douglas Krakower | International Workshop on HIV & Pediatrics 2022 | 27 July 2022
More Study Documents
You must request access to private documents (i.e. call summaries) from the study team. Please email Gabriela Salinas-Jimenez at GSalinas-Jimenez@fhi360.org for access to these documents.
Study Details
To establish the minimum safety, tolerability and acceptability data needed to support the use of cabotegravir long-acting injection (CAB LA) in an adolescent population, potentially transforming the field of HIV prevention for young people.
A Phase IIB single arm, open label safety, tolerability, and acceptability study
Sexually-active, healthy male (assigned at birth) adolescents aged below 18 years
Approximately 50
Participant recruitment will take approximately 12 months. Oral study product will be administered for 5 weeks, followed by 29 weeks on injectable product, then quarterly visits for 48 weeks after final injection. All participants who have received at least one injection will be followed for 48 weeks after their last injection. Waning levels of cabotegravir (the PK tail) will be covered with locally sourced oral TDF/FTC for daily use for 48 weeks. Total study duration per participant will be approximately 21 months.
Step 1 – oral cabotegravir (30mg tablet); Step 2 – 3 mL (600 mg) intramuscular (IM) injection; Step 3 – Tenofovir/Emtricitabine (Trade name: TDF/FTC, Truvada®) (300mg/200mg tablet)
• To evaluate the safety, tolerability and acceptability of CAB LA in healthy HIV-uninfected male adolescents aged below 18 years
• To examine adherence to and timeliness of injections over time among adolescent participants who are provided CAB LA and information regarding its safety and unknown efficacy.
• To examine patterns of sexual risk behavior over time among adolescent participants who are provided CAB LA and information regarding its safety and unknown efficacy.
• To evaluate the safety of CAB LA for 48 weeks of follow-up after final injection.
• To characterize the pharmacokinetics of CAB LA.

