TLC-Plus: A Study to Evaluate the Feasibility of an Enhanced Test, Link to Care, Plus Treat Approach for HIV Prevention in the United States
What is HPTN 065?
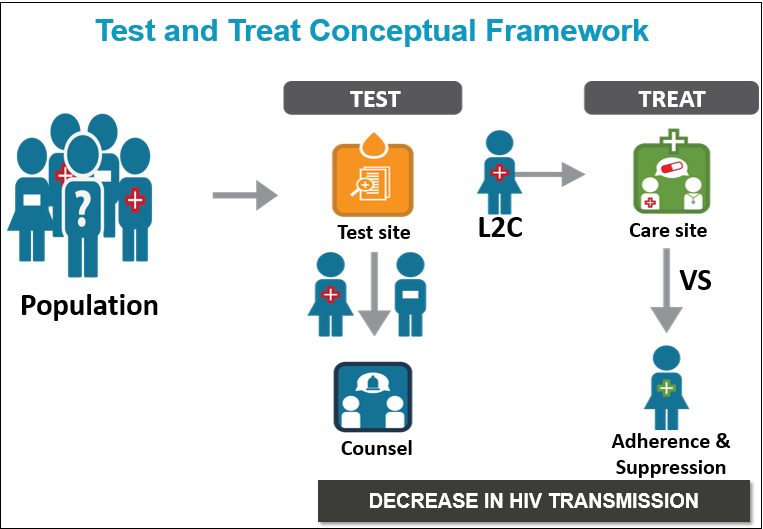
HPTN 065, also known as the Test, Link-to-Care Plus Treat (TLC-Plus) study, was a three-year research study evaluating the feasibility of a community-focused strategy to expand HIV testing, diagnose HIV infection, link HIV-positive individuals to medical care, initiate treatment according to current guidelines and ensure that patients adhere to their treatment regimens. This continuum, which begins with HIV testing and ends with viral suppression, is essential for a successful community-wide strategy centered on the use of antiretroviral therapy for HIV prevention. HPTN 065 (TLC-Plus) was designed with the aim to increase testing, linkage and viral suppression, key steps of the continuum.
Who participated in the study?
The two intervention communities (Bronx, New York and Washington, D.C.) were selected because of their high rates of HIV and they had intensive, ongoing efforts to increase HIV testing and linkage-to-care. In addition, there were four non-intervention communities included in the study— Chicago, Houston, Miami, and Philadelphia—where expanded testing and linkage activities were also underway. Observations in the four non-intervention communities helped assess the influence of changing trends in HIV testing and care expansion in the United States.
What happened during the study?
HPTN 065 consisted of five interrelated study components:
1. Expanded HIV Testing involved social mobilization activities to promote HIV testing in the community and efforts to increase HIV screening of patients seen at participating hospitals in the intervention communities. The goal was to increase the proportion of patients admitted to hospital emergency rooms and inpatient units who were tested for HIV. Activities to expand hospital-based testing took place in 16 participating hospitals (9 in the Bronx and 7 in DC). Social mobilization, which targeted men who have sex with men (MSM) and encouraged HIV testing at least twice a year, took place via various media outlets (print, radio, web, social media) in both DC and the Bronx.
2. Linkage-to-Care used a site-randomized design to evaluate whether financial incentives would increase the likelihood that individuals would be linked with medical care after receiving an HIV diagnosis. A total of 37 HIV testing sites participated in the study (18 in Bronx and 19 in DC). Half of the sites, selected at random (by chance), gave coupons to people who tested positive for HIV. These coupons could be redeemed for $25 and $100 gift cards once they received medical care at HIV care sites. The other half of the test sites continued their usual methods for linking HIV-positive individuals to HIV clinics.
3. Viral Suppression used a site-randomized design to evaluate whether financial incentives would increase the number of patients who take their HIV medication consistently in order to achieve and maintain an undetectable viral load, also referred to as viral suppression. Viral suppression is defined as having an undetectable amount of HIV virus in the blood. A total of 39 HIV care clinics participated in the study (20 in Bronx and 19 in DC). Half of the HIV clinics, selected at random (by chance), gave financial incentives ($70 gift cards every three months) to patients who maintained an undetectable viral load. The other half of the HIV clinics continued their usual methods to help patients take their HIV medications regularly.
4. Prevention for Positives used an individual randomized design to evaluate whether a computerized prevention intervention would reduce risk behaviors, such as unsafe sex or injection drug use more than the standard-of-care prevention services offered by HIV care sites. This study component was conducted at 11 sites (5 in the Bronx and 6 in DC) and enrolled a total of 948 patients.
5. Patient and Provider Surveys assessed the knowledge and attitudes of patients receiving HIV care and HIV care providers about (1) the use of antiretroviral medications for both treatment and prevention of HIV and (2) the use of financial incentives.
HPTN 065 Public Use Data Set
Study Documents
- Financial Incentives Fact Sheet
- Expanded HIV Testing Results Fact Sheet
- Provider Survey Fact Sheet
- Overall Study Fact Sheet
- Community Information Sheet
HPTN 065 Version 3.0
HPTN 065 Version 2.0
- HPTN 065 Protocol V2.0 - 15 Jul 2010
- Summary of Changes to V2.0 - 15 Jul 2010
- LoA #4 - 20 Nov 2012
- LoA #3 - 09 Oct 2012
- LoA #2 - 20 Oct 2011
- LoA #1 - 24 June 2011
- CM 2 - 24 Jan 2013
- CM 1 - 7 Nov 2011
HPTN 065 Version 1.0
Presentations & Webinars
- CROI 2017: Cost-Effectiveness of Financial Incentives for Viral Suppression in HPTN 065
- R4P 2016: Encouraging Trends in HIV Diagnoses, Care and Viral Suppression in 5 US Cities, 2009-2013: Surveillance-based data from HPTN065 (TLC-Plus)
- R4P 2016: Role of Financial Incentives Along the ART Adherence Continuum: A Qualitative Analysis From the HPTN 065 Study
- CROI 2016: Prevention for HIV-infected Persons in HPTN 065: Room for Improvement
- CROI 2016: Computer-based Prevention Counseling for HIV-infected Persons
- CROI 2016: Clinician and Patient Attitudes toward Financial Incentives for HIV Care (HPTN 065)
- NHPC 2015: Evaluation of Process Indicators for Expanded HIV Testing at Hospitals in HPTN 065
- CROI 2015: Expanding HIV Testing in Hospital Emergency Departments and Inpatient Admissions
- CROI 2015: Costs of Expanded HIV Testing in Four Emergency Departments
- CROI 2015: Providers' Attitudes and Practices Related to ART Use for HIV Care and Prevention
- CROI 2015: W. El-Sadr Presentation
- HPTN 065 Qualitative Substudy: The Acceptability of Financial Incentives for Viral Suppression (June 2015)
- R4P 2014: Understanding of Viral Load Among Participants Receiving Financial Incentives for Viral Suppression
- R4P 2014: Acceptability of Financial Incentives for HIV Viral Suppression
- R4P 2014: Geographic Utilization of Gift Cards Used for Financial Incentives to Encourage Viral Suppression
- R4P 2014: The Impact of Implementing a Financial Incentive Program for Viral Suppression on the Clinic Environment
- R4P 2014: Unanticipated Impact of Financial Incentives on HIV Patients and Providers
- AIDS 2012: Community Engagement for the HPTN 065 (TLC-Plus) Study: A Community-Based Study Evaluating the Feasibility of a Combination of Interventions to Prevent HIV Transmission in the US
- AIDS 2012: Clinician Practices and Attitudes Regarding Early Antiretroviral Therapy in the US: Baseline Results from HPTN 065
- AIDS 2012: Linkage-to-Care and ART Adherence Practices at Participating Sites in the HPTN 065 (TLC-Plus) Study
- AIDS 2012: HIV Testing in Six U.S. Cities Using Behavioral Surveillance Data for the TLC-Plus (HPTN 065) Study
- AIDS 2012: Assessing Viral Suppression Amongst HIV Patients Accessing Care in Six Cities Using US HIV Surveillance Data for the TLC-Plus (HPTN 065) Study
Press
Study Details
The main purpose of this study is to assess the feasibility of a community-level test, link to care, plus treat strategy in the United States (U.S.). The study will involve evaluation of the feasibility of some components and the effectiveness of others.
The primary outcomes of the TLC-Plus package of interventions will be determined through measurement of change over the duration of the study in key parameters in two intervention communities. Observations in the four non-intervention control communities will help assess the influence of current trends in HIV testing and care expansion in the U.S.
• TLC-Plus uses innovative approaches, including:
• a community focus
• multi-component strategies that include behavioral and biomedical interventions
• the use of routinely reported HIV surveillance data to determine key outcomes
• a partnership with local Departments of Health and the Centers for Disease Control and Prevention
This study will serve as a proof-of-concept formative study. It will provide key information that could guide the design and anticipate the costs of a future, large, randomized, community-level clinical trial of full implementation of a test-and-treat strategy in the U.S. Findings from this study could also inform test-and-treat efforts in other developed countries with epidemics similar to that in the U.S.
Each component of the study involves an independent design but is interrelated to the other components.
• The Expanded HIV Testing component involves social mobilization, with targeted messaging to promote testing for men who have sex with men (MSM), and implementation of the universal offer of HIV testing in emergency departments (Eds) and hospital inpatient admissions.
• The Linkage-to-Care and Viral Suppression components involve site randomization to test the effectiveness of a financial incentive (FI) intervention compared with the standard of care (SOC).
• The Prevention for Positives component uses individual randomization to compare the SOC plus a computer-delivered intervention with the SOC.
• The Patient and Provider Surveys will be administered at specific time points during the study to assess knowledge, attitudes and practices regarding early initiation of antiretroviral therapy (ART) and the FI interventions.
This study will primarily target individuals age 18 years and older, and will also include younger individuals who are legally able to consent for HIV testing and care according to the state or local law in the two intervention communities. All of the study components will include HIV-positive individuals, with the exception of the provider survey sub-component.
Each of the five study components has a target sample size.
• The universal offer of Expanded HIV Testing will be made in emergency departments and during hospital admission at 16 facilities in the Bronx, NY and Washington, DC. Additionally, focused messages promoting testing for MSM will be targeted to the appropriate population of each intervention community.
• The Linkage-to-Care component includes 37 HIV test sites (18 in the Bronx, NY and 19 in Washington, D.C.) and 39 HIV care sites (20 in the Bronx, NY and 19 in Washington, D.C.). We project that, by the end of the study, approximately 3000 new individuals in the two intervention communities will have tested positive for HIV.
• The Viral Suppression component includes the 39 HIV care sites (20 in the Bronx, NY and 19 in Washington, D.C.). Throughout the study duration, approximately 30,000 HIV-positive individuals will be in care, with an estimated 75% (22,500) eligible for ART in the two intervention communities.
• The Prevention for Positives component will be conducted at a total of twelve sites (six in each intervention community) with up to a total of 1320 patients participating.
• The Patient Survey will be administered to all patients participating in the Prevention for Positives component. Providers at all participating HIV care sites will be invited to complete the provider survey.
The study will take place over 36 months.
The study includes the following five components:
1. Expanded HIV Testing
2. Linkage-to-Care
3. Viral Suppression
4. Prevention for Positives
5. Patient and Provider Surveys
The main objective of the study is to determine the feasibility of a community focused enhanced test and link-to-care strategy in the United States. The study includes feasibility objectives for the Expanded HIV Testing, Linkage-to-Care and Viral Suppression components, and effectiveness objectives for the Linkage-to-Care, Viral Suppression and Prevention for Positive components.